本帖最后由 老马 于 2012-1-13 21:20 编辑
3 p, a: v6 B* _7 u2 x
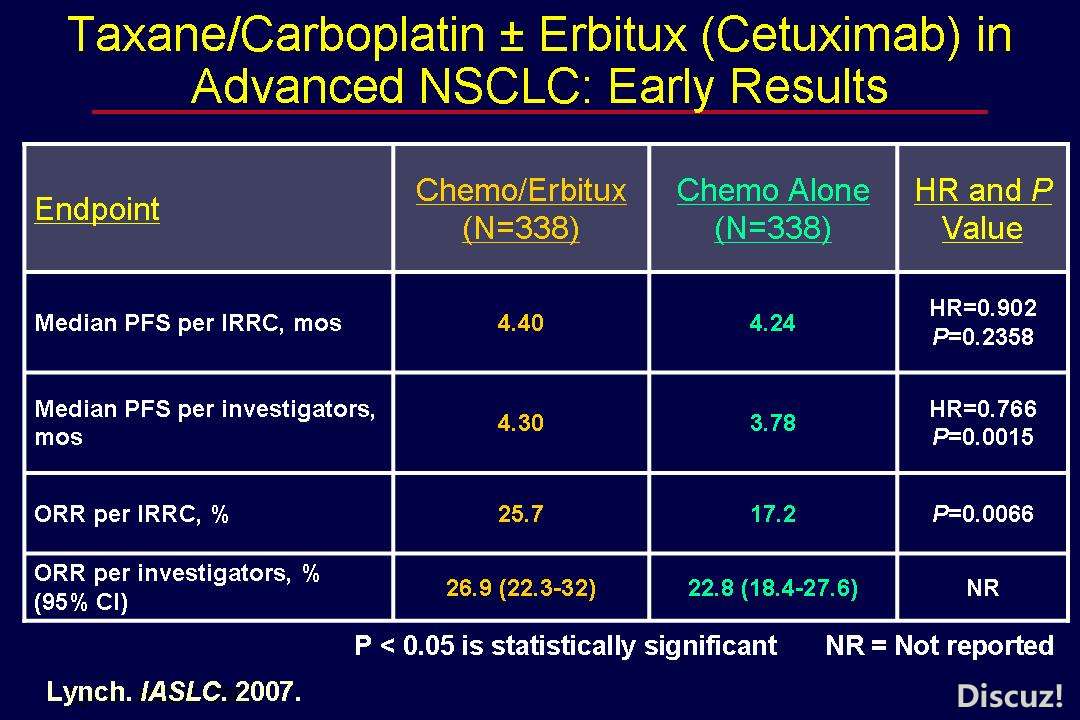
7 D$ E, A. \5 Y5 c* \2 s* p; O6 O1 F爱必妥和阿瓦斯丁的比较
1 j: {6 ]& x- s
 5 s' T' q" t: z, j! B P1 O
5 s' T' q" t: z, j! B P1 O
http://cancergrace.org/lung/2008/08/30/bms099-os-neg/
7 u* A% ?* P% E
 : |) Q; P/ p, z6 f% P
: |) Q; P/ p, z6 f% P
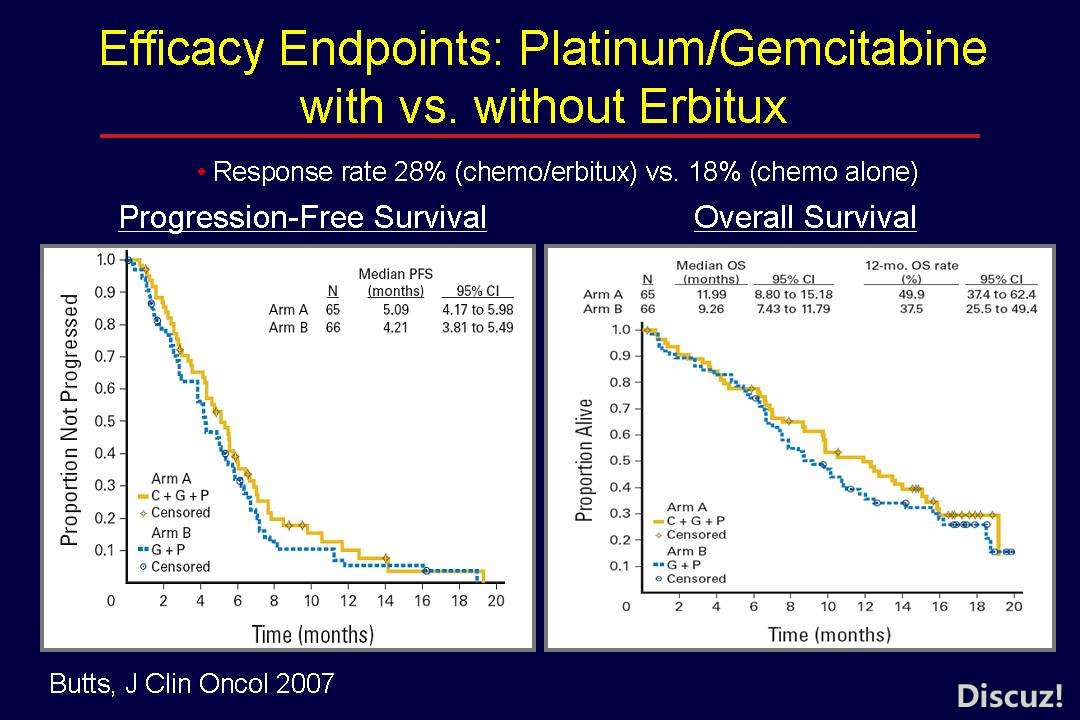
& p6 M/ }5 C" Z1 ~% k0 f$ Jhttp://cancergrace.org/lung/2007/12/27/platgem-erbitux-trial/) @: j# R- x X' y, {, z
==================================================. [4 S0 Q3 K7 a) v2 E% u: S+ u* j
Overall survival with cisplatin–gemcitabine and bevacizumab or placebo as first-line therapy for nonsquamous non-small-cell lung cancer: results from a randomised phase III trial (AVAiL)* _; ~) a$ E; p9 x! D F
Patients and methods: Patients (n = 1043) received cisplatin 80 mg/m2 and gemcitabine 1250 mg/m2 for up to six cycles plus bevacizumab 7.5 mg/kg (n = 345), bevacizumab 15 mg/kg (n = 351) or placebo (n = 347) every 3 weeks until progression. Primary end point was progression-free survival (PFS); OS was a secondary end point.6 N3 a# B4 m+ K9 W
Results: Significant PFS prolongation with bevacizumab compared with placebo was maintained with longer follow-up {hazard ratio (HR) [95% confidence interval (CI)] 0.75 (0.64–0.87), P = 0.0003 and 0.85 (0.73–1.00), P = 0.0456} for the 7.5 and 15 mg/kg groups, respectively. Median OS was >13 months in all treatment groups; nevertheless, OS was not significantly increased with bevacizumab [HR (95% CI) 0.93 (0.78–1.11), P = 0.420 and 1.03 (0.86–1.23), P = 0.761] for the 7.5 and 15 mg/kg groups, respectively, versus placebo. Most patients (~62%) received multiple lines of poststudy treatment. Updated safety results are consistent with those previously reported.- |' ?: ~% a6 p) ^" Q/ m

|